
Rewriting the
Code of Life
How CRISPR Edits a Gene

Rewriting the
Code of Life
How CRISPR Edits a Gene

Rewriting the
Code of Life
Tools that illuminate
your creative process
For most of human history, DNA was something we could only observe. We could map it, study it, and read its instructions, but the idea of reaching into a living cell and changing a single letter in the genetic code belonged to science fiction.
That changed in 2012, when two scientists, Jennifer Doudna and Emmanuelle Charpentier, published a paper that would reshape biology. They had adapted a natural defence system found in bacteria into a tool that could edit genes with extraordinary precision. They called it CRISPR-Cas9.
The discovery earned them the 2020 Nobel Prize in Chemistry and launched a revolution. Within a few years, researchers were using CRISPR to tackle genetic diseases, develop drought-resistant crops, engineer new antibiotics, and even explore the possibility of eliminating malaria-carrying mosquitoes. In 2023, the first CRISPR-based therapy was approved for clinical use in both the UK and the US, treating sickle cell disease and transfusion-dependent beta thalassaemia.
For most of human history, DNA was something we could only observe. We could map it, study it, and read its instructions, but the idea of reaching into a living cell and changing a single letter in the genetic code belonged to science fiction.
That changed in 2012, when two scientists, Jennifer Doudna and Emmanuelle Charpentier, published a paper that would reshape biology. They had adapted a natural defence system found in bacteria into a tool that could edit genes with extraordinary precision. They called it CRISPR-Cas9.
The discovery earned them the 2020 Nobel Prize in Chemistry and launched a revolution. Within a few years, researchers were using CRISPR to tackle genetic diseases, develop drought-resistant crops, engineer new antibiotics, and even explore the possibility of eliminating malaria-carrying mosquitoes. In 2023, the first CRISPR-based therapy was approved for clinical use in both the UK and the US, treating sickle cell disease and transfusion-dependent beta thalassaemia.
For most of human history, DNA was something we could only observe. We could map it, study it, and read its instructions, but the idea of reaching into a living cell and changing a single letter in the genetic code belonged to science fiction.
That changed in 2012, when two scientists, Jennifer Doudna and Emmanuelle Charpentier, published a paper that would reshape biology. They had adapted a natural defence system found in bacteria into a tool that could edit genes with extraordinary precision. They called it CRISPR-Cas9.
The discovery earned them the 2020 Nobel Prize in Chemistry and launched a revolution. Within a few years, researchers were using CRISPR to tackle genetic diseases, develop drought-resistant crops, engineer new antibiotics, and even explore the possibility of eliminating malaria-carrying mosquitoes. In 2023, the first CRISPR-based therapy was approved for clinical use in both the UK and the US, treating sickle cell disease and transfusion-dependent beta thalassaemia.
CRISPR did not invent gene editing, but it made it faster, cheaper, and more accessible than anyone had imagined. A technique that once required years of specialist work could now be carried out in days. Labs that could never have afforded older methods suddenly had the tools to join the effort.
This is the story of how it works,
told at the scale where it happens:
inside a single human cell.
CRISPR did not invent gene editing, but it made it faster, cheaper, and more accessible than anyone had imagined. A technique that once required years of specialist work could now be carried out in days. Labs that could never have afforded older methods suddenly had the tools to join the effort.
This is the story of how it works,
told at the scale where it happens:
inside a single human cell.
CRISPR did not invent gene editing, but it made it faster, cheaper, and more accessible than anyone had imagined. A technique that once required years of specialist work could now be carried out in days. Labs that could never have afforded older methods suddenly had the tools to join the effort.
This is the story of how it works,
told at the scale where it happens:
inside a single human cell.
Inside the Cell
Every human cell contains roughly two metres of DNA, coiled and folded into a space just a few thousandths of a millimetre across. That DNA holds the complete set of instructions needed to build and maintain a human body: around 20,000 genes encoded in over three billion pairs of chemical letters.
To reach a gene, you first have to get inside the cell. The outer boundary is the cell membrane, a double-layered wall of fat molecules studded with proteins that act as gatekeepers, letting some things in and keeping others out. Beyond the membrane lies the cytoplasm, a dense, gel-like interior filled with organelles. Mitochondria generate the cell’s energy. The endoplasmic reticulum folds and transports proteins. And the Golgi apparatus packages molecules for delivery. Each one is a miniature factory with its own job to do.
At the centre sits the nucleus, the cell’s command centre, enclosed in its own double membrane and peppered with ring-shaped pores that control what passes in and out. Inside the nucleus, DNA is not the neat, bare double helix you see in textbooks. It exists as chromatin: a dense, tangled mass of fibres, with the DNA strand wrapped around tiny spool-shaped proteins called histones, like thread wound around beads. Scientists describe the overall structure as a “polymer melt”: dynamic, shifting, and irregular.
Somewhere in that tangle is the gene that CRISPR has been designed to find.
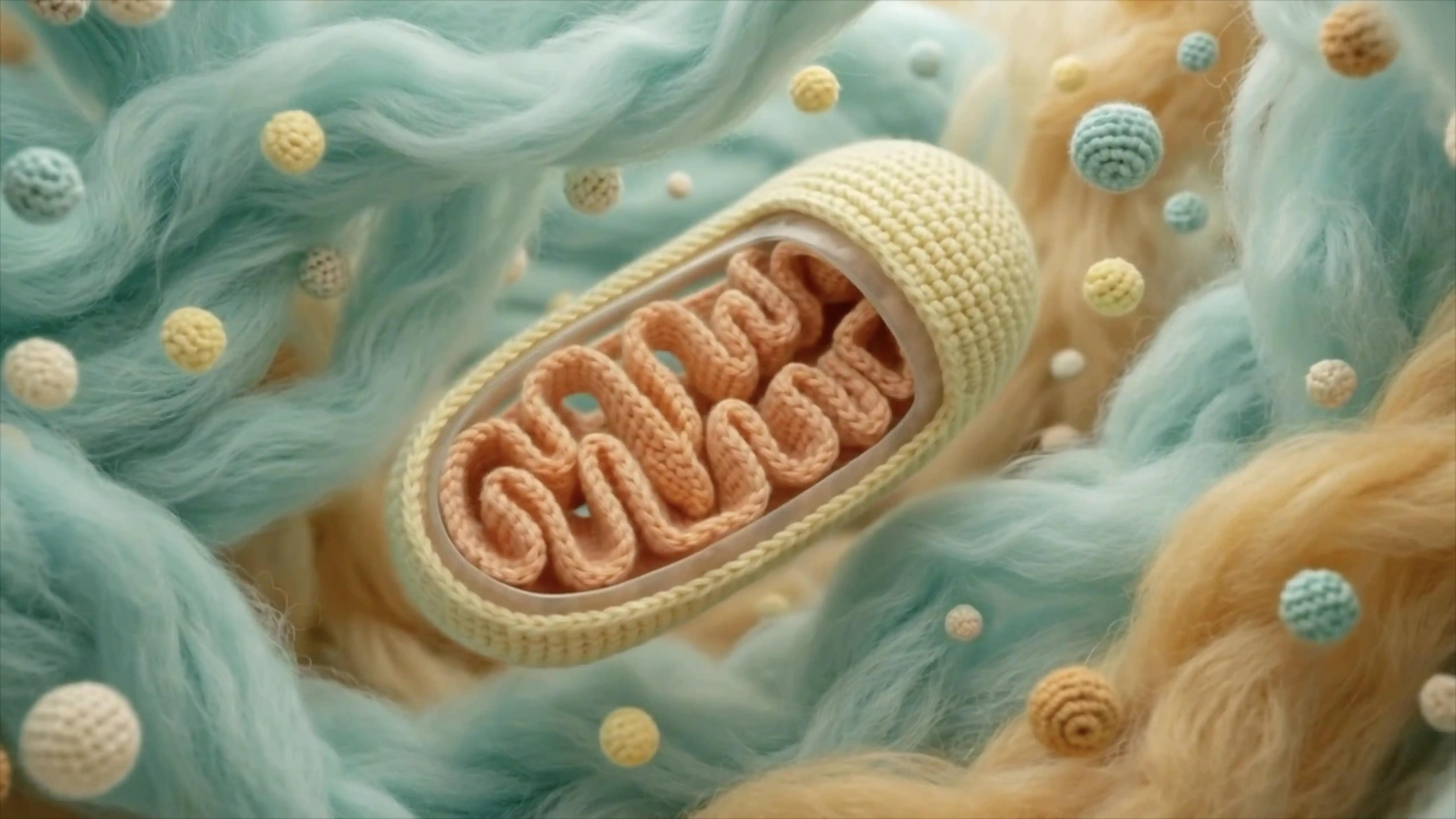
Inside the Cell
Every human cell contains roughly two metres of DNA, coiled and folded into a space just a few thousandths of a millimetre across. That DNA holds the complete set of instructions needed to build and maintain a human body: around 20,000 genes encoded in over three billion pairs of chemical letters.
To reach a gene, you first have to get inside the cell. The outer boundary is the cell membrane, a double-layered wall of fat molecules studded with proteins that act as gatekeepers, letting some things in and keeping others out. Beyond the membrane lies the cytoplasm, a dense, gel-like interior filled with organelles. Mitochondria generate the cell’s energy. The endoplasmic reticulum folds and transports proteins. And the Golgi apparatus packages molecules for delivery. Each one is a miniature factory with its own job to do.
At the centre sits the nucleus, the cell’s command centre, enclosed in its own double membrane and peppered with ring-shaped pores that control what passes in and out. Inside the nucleus, DNA is not the neat, bare double helix you see in textbooks. It exists as chromatin: a dense, tangled mass of fibres, with the DNA strand wrapped around tiny spool-shaped proteins called histones, like thread wound around beads. Scientists describe the overall structure as a “polymer melt”: dynamic, shifting, and irregular.
Somewhere in that tangle is the gene that CRISPR has been designed to find.

Inside the Cell
Every human cell contains roughly two metres of DNA, coiled and folded into a space just a few thousandths of a millimetre across. That DNA holds the complete set of instructions needed to build and maintain a human body: around 20,000 genes encoded in over three billion pairs of chemical letters.
To reach a gene, you first have to get inside the cell. The outer boundary is the cell membrane, a double-layered wall of fat molecules studded with proteins that act as gatekeepers, letting some things in and keeping others out. Beyond the membrane lies the cytoplasm, a dense, gel-like interior filled with organelles. Mitochondria generate the cell’s energy. The endoplasmic reticulum folds and transports proteins. And the Golgi apparatus packages molecules for delivery. Each one is a miniature factory with its own job to do.
At the centre sits the nucleus, the cell’s command centre, enclosed in its own double membrane and peppered with ring-shaped pores that control what passes in and out. Inside the nucleus, DNA is not the neat, bare double helix you see in textbooks. It exists as chromatin: a dense, tangled mass of fibres, with the DNA strand wrapped around tiny spool-shaped proteins called histones, like thread wound around beads. Scientists describe the overall structure as a “polymer melt”: dynamic, shifting, and irregular.
Somewhere in that tangle is the gene that CRISPR has been designed to find.


Finding the Target
CRISPR gene editing relies on two key components working together: a protein called Cas9 and a short piece of synthetic RNA known as the guide RNA.
Cas9 is the cutting tool. Under a powerful enough microscope, it looks like a lumpy, two-lobed blob, roughly 10 nanometres across. One lobe (the recognition lobe) holds the guide RNA. The other (the nuclease lobe) contains two molecular blades that will eventually make the cut. Between them sits a central channel where the DNA will be read. When the guide RNA loads in, the two lobes swing apart, opening that channel and priming the protein for its search.
The guide RNA is the satnav. It is a single strand of RNA, about 100 nucleotides long, with a crucial 20-letter sequence at one end. Those 20 letters are custom-designed by researchers to match one specific location in the genome. The rest of the RNA forms a scaffold that locks it into position inside Cas9.
Together, Cas9 and its guide drift along the DNA, scanning for a match. The process is surprisingly physical. Cas9 slides along the strand, pausing every time it encounters a short three-letter code called a PAM sequence. At each PAM, it briefly pries open the double helix to check whether the adjacent sequence matches its guide. If there is no match, it lets go and moves on. It has been compared to someone flipping through the pages of a book, looking for a specific passage.
Cas9 may check hundreds or even thousands of sites before it finds the right one.

Finding the Target
CRISPR gene editing relies on two key components working together: a protein called Cas9 and a short piece of synthetic RNA known as the guide RNA.
Cas9 is the cutting tool. Under a powerful enough microscope, it looks like a lumpy, two-lobed blob, roughly 10 nanometres across. One lobe (the recognition lobe) holds the guide RNA. The other (the nuclease lobe) contains two molecular blades that will eventually make the cut. Between them sits a central channel where the DNA will be read. When the guide RNA loads in, the two lobes swing apart, opening that channel and priming the protein for its search.
The guide RNA is the satnav. It is a single strand of RNA, about 100 nucleotides long, with a crucial 20-letter sequence at one end. Those 20 letters are custom-designed by researchers to match one specific location in the genome. The rest of the RNA forms a scaffold that locks it into position inside Cas9.
Together, Cas9 and its guide drift along the DNA, scanning for a match. The process is surprisingly physical. Cas9 slides along the strand, pausing every time it encounters a short three-letter code called a PAM sequence. At each PAM, it briefly pries open the double helix to check whether the adjacent sequence matches its guide. If there is no match, it lets go and moves on. It has been compared to someone flipping through the pages of a book, looking for a specific passage.
Cas9 may check hundreds or even thousands of sites before it finds the right one.

Finding the Target
CRISPR gene editing relies on two key components working together: a protein called Cas9 and a short piece of synthetic RNA known as the guide RNA.
Cas9 is the cutting tool. Under a powerful enough microscope, it looks like a lumpy, two-lobed blob, roughly 10 nanometres across. One lobe (the recognition lobe) holds the guide RNA. The other (the nuclease lobe) contains two molecular blades that will eventually make the cut. Between them sits a central channel where the DNA will be read. When the guide RNA loads in, the two lobes swing apart, opening that channel and priming the protein for its search.
The guide RNA is the satnav. It is a single strand of RNA, about 100 nucleotides long, with a crucial 20-letter sequence at one end. Those 20 letters are custom-designed by researchers to match one specific location in the genome. The rest of the RNA forms a scaffold that locks it into position inside Cas9.
Together, Cas9 and its guide drift along the DNA, scanning for a match. The process is surprisingly physical. Cas9 slides along the strand, pausing every time it encounters a short three-letter code called a PAM sequence. At each PAM, it briefly pries open the double helix to check whether the adjacent sequence matches its guide. If there is no match, it lets go and moves on. It has been compared to someone flipping through the pages of a book, looking for a specific passage.
Cas9 may check hundreds or even thousands of sites before it finds the right one.
MANIFESTO
The Lock & the Cut
When Cas9 finally lands on the correct sequence, the guide RNA begins forming base pairs with one strand of the DNA, letter by letter, starting from the PAM end and working along the sequence. As the RNA binds, it pushes the opposite DNA strand aside, creating a bubble-like structure called an R-loop. The double helix unwinds locally, and the match is confirmed.
At that point, Cas9 shifts shape. An internal component called the HNH domain swings into position over the target strand, closing a gap of about 2.5 nanometres in a dramatic hinge-like motion. The protein tightens its grip. It is now armed.
Then it cuts. Cas9 has two separate blades, each responsible for severing one strand of the double helix. The HNH domain cuts the strand paired with the guide RNA. The RuvC domain cuts the opposite strand. Both cuts happen at almost the same position, producing a clean break through both strands of the DNA. The two severed ends drift apart, and the genetic code is now open.

MANIFESTO
The Lock & the Cut
When Cas9 finally lands on the correct sequence, the guide RNA begins forming base pairs with one strand of the DNA, letter by letter, starting from the PAM end and working along the sequence. As the RNA binds, it pushes the opposite DNA strand aside, creating a bubble-like structure called an R-loop. The double helix unwinds locally, and the match is confirmed.
At that point, Cas9 shifts shape. An internal component called the HNH domain swings into position over the target strand, closing a gap of about 2.5 nanometres in a dramatic hinge-like motion. The protein tightens its grip. It is now armed.
Then it cuts. Cas9 has two separate blades, each responsible for severing one strand of the double helix. The HNH domain cuts the strand paired with the guide RNA. The RuvC domain cuts the opposite strand. Both cuts happen at almost the same position, producing a clean break through both strands of the DNA. The two severed ends drift apart, and the genetic code is now open.

MANIFESTO
The Lock & the Cut
When Cas9 finally lands on the correct sequence, the guide RNA begins forming base pairs with one strand of the DNA, letter by letter, starting from the PAM end and working along the sequence. As the RNA binds, it pushes the opposite DNA strand aside, creating a bubble-like structure called an R-loop. The double helix unwinds locally, and the match is confirmed.
At that point, Cas9 shifts shape. An internal component called the HNH domain swings into position over the target strand, closing a gap of about 2.5 nanometres in a dramatic hinge-like motion. The protein tightens its grip. It is now armed.
Then it cuts. Cas9 has two separate blades, each responsible for severing one strand of the double helix. The HNH domain cuts the strand paired with the guide RNA. The RuvC domain cuts the opposite strand. Both cuts happen at almost the same position, producing a clean break through both strands of the DNA. The two severed ends drift apart, and the genetic code is now open.

Writing the Fix
A double-strand break in DNA is a serious event. The cell recognises it immediately and sends in its repair machinery: a cluster of proteins that converge on the break site like a maintenance crew arriving at a fault line.
Left to its own devices, the cell would simply glue the two broken ends back together, a quick-fix process called non-homologous end joining. This is useful for disabling a gene (the repair is often imperfect, introducing small errors that stop the gene from working), but it is not precise enough for making a specific correction.
For precision editing, researchers provide something extra alongside Cas9: a donor template. This is a short piece of synthetic DNA carrying the corrected or new sequence, flanked by stretches that match the DNA on either side of the cut. The cell’s repair machinery reads this template like a blueprint and copies the new information into the gap. The result is a seamless edit: one sequence swapped for another, permanently written into the cell’s genome.
As the repair completes, the double helix rejoins. The DNA re-coils into its chromatin packing, and the edited gene takes its place in the genome, ready to be read by the cell.

Writing the Fix
A double-strand break in DNA is a serious event. The cell recognises it immediately and sends in its repair machinery: a cluster of proteins that converge on the break site like a maintenance crew arriving at a fault line.
Left to its own devices, the cell would simply glue the two broken ends back together, a quick-fix process called non-homologous end joining. This is useful for disabling a gene (the repair is often imperfect, introducing small errors that stop the gene from working), but it is not precise enough for making a specific correction.
For precision editing, researchers provide something extra alongside Cas9: a donor template. This is a short piece of synthetic DNA carrying the corrected or new sequence, flanked by stretches that match the DNA on either side of the cut. The cell’s repair machinery reads this template like a blueprint and copies the new information into the gap. The result is a seamless edit: one sequence swapped for another, permanently written into the cell’s genome.
As the repair completes, the double helix rejoins. The DNA re-coils into its chromatin packing, and the edited gene takes its place in the genome, ready to be read by the cell.

Writing the Fix
A double-strand break in DNA is a serious event. The cell recognises it immediately and sends in its repair machinery: a cluster of proteins that converge on the break site like a maintenance crew arriving at a fault line.
Left to its own devices, the cell would simply glue the two broken ends back together, a quick-fix process called non-homologous end joining. This is useful for disabling a gene (the repair is often imperfect, introducing small errors that stop the gene from working), but it is not precise enough for making a specific correction.
For precision editing, researchers provide something extra alongside Cas9: a donor template. This is a short piece of synthetic DNA carrying the corrected or new sequence, flanked by stretches that match the DNA on either side of the cut. The cell’s repair machinery reads this template like a blueprint and copies the new information into the gap. The result is a seamless edit: one sequence swapped for another, permanently written into the cell’s genome.
As the repair completes, the double helix rejoins. The DNA re-coils into its chromatin packing, and the edited gene takes its place in the genome, ready to be read by the cell.

What CRISPR
Makes Possible
The implications of being able to edit genes
this precisely are enormous, and they
are already being realised.

In medicine, CRISPR-based therapies have moved from laboratory experiments to approved treatments. Casgevy, the first CRISPR therapy to receive regulatory approval, treats sickle cell disease and beta thalassaemia by editing a patient’s own blood stem cells. Clinical trials are exploring CRISPR-based approaches to cancers, inherited blindness, high cholesterol, and HIV.
In agriculture, gene-edited crops are being developed that can withstand drought, resist disease, and grow with fewer pesticides. Unlike older methods of genetic modification, CRISPR can make targeted changes without introducing DNA from other species, which has shifted regulatory and public conversations around the technology.
In basic research, CRISPR has become an everyday tool. Scientists use it to study gene function, model diseases in the laboratory, and investigate everything from antibiotic resistance to the biology of ageing.
The technology continues to evolve. Newer variants like base editing and prime editing can change individual DNA letters or insert new sequences without making a double-strand break at all, offering even greater precision and fewer unintended changes.
What CRISPR
Makes Possible
The implications of being able to edit genes
this precisely are enormous, and they
are already being realised.

In medicine, CRISPR-based therapies have moved from laboratory experiments to approved treatments. Casgevy, the first CRISPR therapy to receive regulatory approval, treats sickle cell disease and beta thalassaemia by editing a patient’s own blood stem cells. Clinical trials are exploring CRISPR-based approaches to cancers, inherited blindness, high cholesterol, and HIV.
In agriculture, gene-edited crops are being developed that can withstand drought, resist disease, and grow with fewer pesticides. Unlike older methods of genetic modification, CRISPR can make targeted changes without introducing DNA from other species, which has shifted regulatory and public conversations around the technology.
In basic research, CRISPR has become an everyday tool. Scientists use it to study gene function, model diseases in the laboratory, and investigate everything from antibiotic resistance to the biology of ageing.
The technology continues to evolve. Newer variants like base editing and prime editing can change individual DNA letters or insert new sequences without making a double-strand break at all, offering even greater precision and fewer unintended changes.
What CRISPR
Makes Possible
The implications of being able to edit genes this precisely are enormous, and they are already being realised.

In medicine, CRISPR-based therapies have moved from laboratory experiments to approved treatments. Casgevy, the first CRISPR therapy to receive regulatory approval, treats sickle cell disease and beta thalassaemia by editing a patient’s own blood stem cells. Clinical trials are exploring CRISPR-based approaches to cancers, inherited blindness, high cholesterol, and HIV.
In agriculture, gene-edited crops are being developed that can withstand drought, resist disease, and grow with fewer pesticides. Unlike older methods of genetic modification, CRISPR can make targeted changes without introducing DNA from other species, which has shifted regulatory and public conversations around the technology.
In basic research, CRISPR has become an everyday tool. Scientists use it to study gene function, model diseases in the laboratory, and investigate everything from antibiotic resistance to the biology of ageing.
The technology continues to evolve. Newer variants like base editing and prime editing can change individual DNA letters or insert new sequences without making a double-strand break at all, offering even greater precision and fewer unintended changes.
The Bigger Picture
CRISPR raises questions as big as its possibilities. The ability to rewrite genetic code brings with it profound ethical debates about where the boundaries should lie.

In 2018, a Chinese scientist announced he had used CRISPR to edit the genes of human embryos, resulting in the birth of twin girls. A third baby was born from a separate pregnancy the following year. The experiments were widely condemned by the scientific community, as they were carried out without proper oversight, with forged ethics documents, and misleading consent processes. Furthermore, the long-term consequences for the children remain unknown. The researcher was sentenced to three years in prison, and the case underscored the difference between editing the genes of a patient (changes that affect only that individual) and editing embryos or reproductive cells (changes that would be inherited by future generations).
Most scientists and ethicists agree that somatic gene editing, targeting cells in a living patient, holds tremendous therapeutic promise and is subject to the same regulatory frameworks as other medical treatments. Germline editing, which alters the DNA passed to future generations, remains far more contentious. In 2019, the World Health Organization’s Director-General stated that it would be irresponsible for anyone to proceed with clinical applications of human germline genome editing, and the WHO has since published a governance framework and recommendations to guide the responsible development of the technology.
The Bigger Picture
CRISPR raises questions as big as its possibilities. The ability to rewrite genetic code brings with it profound ethical debates about where the boundaries should lie.

In 2018, a Chinese scientist announced he had used CRISPR to edit the genes of human embryos, resulting in the birth of twin girls. A third baby was born from a separate pregnancy the following year. The experiments were widely condemned by the scientific community, as they were carried out without proper oversight, with forged ethics documents, and misleading consent processes. Furthermore, the long-term consequences for the children remain unknown. The researcher was sentenced to three years in prison, and the case underscored the difference between editing the genes of a patient (changes that affect only that individual) and editing embryos or reproductive cells (changes that would be inherited by future generations).
Most scientists and ethicists agree that somatic gene editing, targeting cells in a living patient, holds tremendous therapeutic promise and is subject to the same regulatory frameworks as other medical treatments. Germline editing, which alters the DNA passed to future generations, remains far more contentious. In 2019, the World Health Organization’s Director-General stated that it would be irresponsible for anyone to proceed with clinical applications of human germline genome editing, and the WHO has since published a governance framework and recommendations to guide the responsible development of the technology.
The Bigger Picture
CRISPR raises questions as big as its possibilities. The ability to rewrite genetic code brings with it profound ethical debates about where the boundaries should lie.

In 2018, a Chinese scientist announced he had used CRISPR to edit the genes of human embryos, resulting in the birth of twin girls. A third baby was born from a separate pregnancy the following year. The experiments were widely condemned by the scientific community, as they were carried out without proper oversight, with forged ethics documents, and misleading consent processes. Furthermore, the long-term consequences for the children remain unknown. The researcher was sentenced to three years in prison, and the case underscored the difference between editing the genes of a patient (changes that affect only that individual) and editing embryos or reproductive cells (changes that would be inherited by future generations).
Most scientists and ethicists agree that somatic gene editing, targeting cells in a living patient, holds tremendous therapeutic promise and is subject to the same regulatory frameworks as other medical treatments. Germline editing, which alters the DNA passed to future generations, remains far more contentious. In 2019, the World Health Organization’s Director-General stated that it would be irresponsible for anyone to proceed with clinical applications of human germline genome editing, and the WHO has since published a governance framework and recommendations to guide the responsible development of the technology.

These are not distant hypotheticals
As the technology becomes cheaper and more widely available, the conversations around access, consent, equity, and oversight are becoming more urgent. Who gets to benefit from gene editing? Who decides which conditions are worth correcting? How do we ensure the technology is used responsibly?
There are no simple answers, but the questions matter. CRISPR has given us an extraordinary capability. What we choose to do with it will define a chapter of human history.

These are not distant hypotheticals
As the technology becomes cheaper and more widely available, the conversations around access, consent, equity, and oversight are becoming more urgent. Who gets to benefit from gene editing? Who decides which conditions are worth correcting? How do we ensure the technology is used responsibly?
There are no simple answers, but the questions matter. CRISPR has given us an extraordinary capability. What we choose to do with it will define a chapter of human history.

These are not distant hypotheticals
As the technology becomes cheaper and more widely available, the conversations around access, consent, equity, and oversight are becoming more urgent. Who gets to benefit from gene editing? Who decides which conditions are worth correcting? How do we ensure the technology is used responsibly?
There are no simple answers, but the questions matter. CRISPR has given us an extraordinary capability. What we choose to do with it will define a chapter of human history.